The reasons for measuring the spectroscopic properties of biological systems are many. Native or stained tissue and cells absorb and emit light in such a way as to leave distinct spectral signatures. Spectroscopy provides us a means to directly identify and quantify substances by way of this unique fingerprint spectrum, and can be thought of as a type of "chemical vision". By measuring subtle changes to the spectral profiles of histological and cytological samples for example, or even of in vivo tissue through use of fibre-optic probes, one has a means of determining the onset and nature of neoplasia. Spectroscopic detection can also be used to examine retinal disease states. In fluorescence microscopy, fixed or living cells are often labelled with multiple fluorescent probes, each specific to a particular intracellular component. The challenge then becomes - how do we separate the various distinct, but often spectrally overlapping, signals from this battery of fluorescent labels to enable us to build up an accurate, high-contrast and quantitative representation of the location, concentration and interactions of the intracellular components under investigation? This is an important and justifiable challenge, as in the post-genomic era, modern techniques involving multi-labeling are revolutionising how we directly probe and understand protein function and key physiological mechanisms of the cell, and associated disease states.
Finally Raman spectroscopy, while being an inherently weak phenomenon, carries the considerable advantage that Raman bands have very narrow bandwidths compared to typical fluorescence bands. As such, Raman is ideal for spectral discrimination of multiple spatially overlapped species, all the more powerful when combined with deconvolution and multi-variate analysis.
The concept of spectral imaging - generating quantitative 2D spatial images over a third spectral dimension to yield a "data cube" - has been originally substantiated within fields such as airborne surveillance or satellite imaging, in order to generate spectroscopic data detailing the chemical composition of test areas. Such a configuration might involve focusing a line of incoming signal into an imaging spectrograph where the resulting spectrally dispersed light from that line is cast over an area of the imaging CCD. The airborne craft, and therefore the attached detection system, is "scanned" over the surface of the ground with the CCD operating at a sufficiently fast frame rate, and 3D information is rapidly built up. The data cube can be viewed in two principle ways: a) one can derive a spectrum from any spatial point on the image
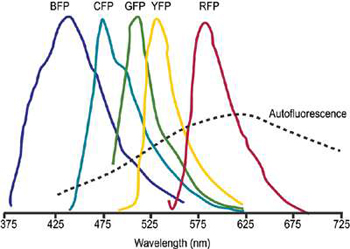
Schematic representation of overlapped fluorescent protein emission spectra. Typicallybroad autofluorescence is shown also.
b) one can generate a series of discrete single wavelength images. When the spectrum is dispersed over many spectral channels (or pixels in the case of using CCDs), the technique is often called hyper- or multi-spectral imaging. Development of the spectral imaging approach within areas such as surveillance has been continuing for over 20 years, and much of the information learned can be carried over to help solve biological problems.
Within fluorescence microscopy, particularly on living cells, the concept of multi-spectral imaging is being embraced as a means to collect full and detailed dynamic information from extensively labelled samples. Quantitative spectral "unmixing" algorithms (of which a number exist) can be applied to deconvolve the spectra from each image point into its constituent emitting species and their relative contribution. Importantly, unmixing can be successfully applied to cases where there is significant spectral overlapping of multiple dyes. This facilitates use of fluorescent proteins such as GFP and its most common mutations, CFP, YFP and ds-RED, in techniques such as co-localization and FRET. FRET, by its very nature, is particularly prone to significant spectral "cross-talk". For efficient FRET, there needs to be a significant degree of overlap between the emission spectrum of the donor dye and the absorption spectrum of the acceptor. This condition is often inextricably accompanied by considerable overlie of the two emission spectra, as is the case for the common FRET pair GFP/YFP.
It is possible to carry out successful FRET using a combination of narrow band filters, and split the spectrally distinct images onto either two cameras or adjacent fields of view within the same camera (up to four fields can be achieved at present). Excitation of both donor and acceptor species, and subsequent unmixing from as little as two "filter-based channels" can be executed to minimize effects of cross-talk. This method has the advantage of affording rapid and simultaneous imaging of two or three fluorophores. Where we deal with a greater number of fluorophores however, and especially when they are significantly spectrally overlapped, cross-talk becomes much more of an issue and we require more spectral channels to effectively separate the signals. It is conceivable however, that rather that using narrow band filters to separate signal from closely overlapping fluorophores, we instead perform rapid multi-spectral imaging and collect the full unfiltered emission spectra, followed by unmixing. If this configuration can be realised, better signal-to-noise images are obtained as we do not have to "throw away" the majority of emitted photons through use of narrow band filters. Furthermore, through spectral imaging and unmixing, we can also separate out and remove any autofluorescence from the sample.
However, while the benefits of such an approach to multi-spectral image separation are substantial, as too are the technical challenges. The ideal spectral imaging system would have a number of key attributes. It would be capable of rapidly collecting spectral data for generation of data cubes with maximum speed, facilitating both throughput and study of dynamic samples. The system would be sufficiently sensitive in terms of high optical throughput, minimal detector noise and maximum detector Quantum Efficiency (QE), to yielding a high signal-to-noise ratio – it has been shown that efficacy of spectral unmixing is substantially greater under such conditions. Spectral resolution should be sufficient as to enable effective separation of the constituent contributing species – the number of spectral channels practically required is driven by: a) the number of contributing overlapped species and
b) whether or not we can use existing reference emission spectra for unmixing (or whether we are recording "unknowns"). As mentioned previously above, if the method involves collecting the entire overlapped spectrum over the wavelength range and therefore avoiding use of narrow band filters as the detection channels, one does not have to waste photons in achieving the desired spectral resolution and separation, resulting in higher Signal-to-Noise (S/N), better resolution and more accurate quantification.
Andor's EMCCD technology is ideal for incorporation into your ultra-sensitive spectral imaging set-up. The extraordinary S/N offered is significantly greater than that afforded by conventional CCD cameras operating at fast readout speeds. Furthermore, since it is a fast-readout imaging device the EMCCD yields frame rates that are ideally suited to the likes of dynamic FRET events or recording of faster data cubes. Underlying all direct imaging studies of living cells, is the desire to preserve the living subject for as long as possible, through minimization of both phototoxic cell/tissue damage and also photo-bleaching of the incorporated fluorophores. As such, the techniques benefit markedly from using EMCCD detection technology, which not only exhibits the sensitivity and speed to capture, with high S/N, spectrally resolved image sets from weakly emitting species, but also enables the laser excitation power to be attenuated. Through minimization of excitation powers, the rates of dye photo-bleaching and cell phototoxicity are significantly reduced.
The traditional way of spectroscopically resolving wavelengths with CCDs, has been to use a dispersive element such as a grating, which will cast the wavelength separated light across one dimension of the array. Indeed, dedicated spectroscopic CCD formats exist specifically for this usage – these tend to be elongated in shape, typical pixel formats being 1024 x 128 or 1600 x 200. In standard spectroscopy readout mode, the light is dispersed along the longer horizontal dimension giving > 1000 "spectral channels" and the vertical columns are binned on chip (full vertical binning), creating a larger light collection area and speeding up the obtainable spectral frame rate.
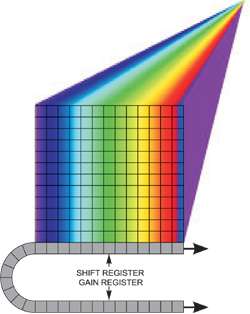
Spectral dispersion from a line illumination
The high-end iXon3 EMCCD cameras that were pioneered by Andor, contain imaging sensor formats such as 512 x 512 or 1000 x 1000. These have been adapted for use in standard spectroscopic mode, performing extensive vertical binning along the vertical column dimension. However, a dedicated spectroscopy EMCCD camera has recently been pioneered, Andor's NewtonEM EMCCD camera. This USB 2.0, -90°C Thermoelectric (TE) cooled platform is available in either 1600 x 256 or 1600 x 128 pixel formats, and is optimally suited to rapid low-light spectroscopic applications.
Other useful ways of reading out EMCCDs spectroscopically are commonly called upon. For example, it is possible to make use of an imaging spectrograph, such as Andor's Shamrock 303i, that will provide spatial resolution along the dimension of the entrance slit.The EMCCD can be read out as a complete image, with spectral resolution along the horizontal axis and the vertical spatial dimension perpendicular to this. The sensor can even be read out in multiple horizontal tracks down the array, of software-configurable size and position, each one giving a separate spectrum, corresponding for example to the input of an optical fibre from a point in the sample.
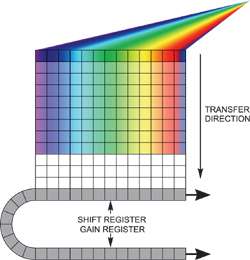
Fast kinetics readout
If we need to achieve extraordinarily fast spectral rates, two options are available, but for each we must be able to ensure that light is dispersed only over a limited number of rows on the sensor with no light falling onto the remainder of the active area. The fastest time resolution is obtained by reading out an imaging EMCCD in "fast kinetics mode". In this configuration, the spectrum is dispersed over a few rows at the very top of the sensor. The "dark rows" beneath are subsequently used for storing the spectra shifted down from the exposed area, this continuing until the entire sensor is filled – the whole area is then read out as normal, and the software processes the multiple spectra. Thus one can achieve time resolution down to the time taken to shift these few rows down by one step, which in the Andor iXonEM+ EMCCD range is tuneable to < 1 μs/row, with up to 1999 storage rows.
If your experiment dictates that you need both fast spectral time resolution but cannot be constrained by the storage size of the sensor, then it is feasible to readout the EMCCD in a "cropped sensor" mode. In this mode, we can "fool" the sensor into thinking it is smaller than it actually is, and readout continuously at a much faster frame rate! The spectral time resolution is dictated by the time taken to readout the smaller defined section of the sensor. For example, by focusing dispersed light onto a 10 x 1000 area at the bottom of iXonEM+ DU885-K at 35 MHz pixel readout, one can then readout at 10's of thousands of spectra/sec.
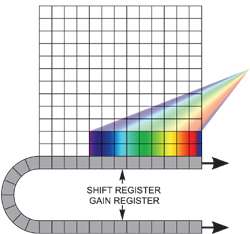
Cropped sensor mode
A number of novel spectral imaging techniques have been implemented to date that harness the rapid readout and ultrasensitive characteristics of EMCCDs.
An area that is being addressed is that of next-generation multi-spectral confocal scanning. A number of commercial systems for multi-spectral confocal point scanning microscopy exist, but with some considerable drawbacks. Two formats in particular are worth mentioning. One configuration involves employing a grating to disperse the confocal signal from each scanned point over a linear array of 32 Photomultiplier Tubes (PMTs). This suffers primarily from the notably low QE of PMTs (especially towards the red) and diverse responses of neighboring channels. Another current commercial format employs a single PMT for detection, and tilt-scans a grating across the spectral range during the dwell-time of laser on an image point. It is not hard to conceive that it takes a considerable time to perform spectral scans on > 250,000 pixels of a 512 x 512 image, and such a configuration is not well suited to spectral study of dynamic systems, as well as suffering the adverse effects of extensive sample illumination.
To overcome these performance shortcomings, it is possible to implement EMCCDs system for multi-spectral confocal scanning. The > 90% quantum efficiency of the back-illuminated sensor, single photon sensitivity, array architecture and rapid pixel readout speed is exploited to markedly improve this approach. The laser dwell-time should be set to coincide with the time to expose and read-out a short row of approx 32 pixels - sufficient spectral channels to yield effective unmixing of several known emitting dyes, resulting in a data cube of 512 x 512 x 32 taking less than 1 second to generate (for the 10 MHz of the iXonEM+ DU887 – faster rates available from the 35MHZ DV-885). Faster speeds still can be expected with fewer spectral channels. There is a clear sensitivity advantage of EMCCD pixels over the usually employed PMT-technology, which is circa 5-fold in the blue-green and up to tenfold in the red.
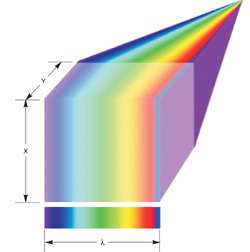 Line scan spectral imaging
Line scan spectral imaging
It is possible also to perform multi-spectral imaging in microscopy through making use of modules that contain transmissive dispersion gratings, such as the Imspector. The optical principle of dispersion and detection in these systems is somewhat analogous to that described earlier for airborne monitoring. This unit is mounted between microscope and EMCCD and light is collected from an illuminated line in the x-direction across the sample – line illumination coming either from filtered lamp or laser. The collected light from the line is dispersed over the area of the EMCCD array, or even part of the available area for faster frame rates with lower spectral range or resolution. Thus the pixels across the columns of the EMCCD provide resolution in λ. The y-direction is scanned through moving the sample incrementally via a motorized stage. For every stage step (typically 512), one needs to expose and readout the area of the array that has been illuminated. The ultra-sensitive performance of EMCCDs is ideal for this approach, enabling both shorter exposure times and faster readout performance.
EMCCD performance can readily be applied to speed up a spectral imaging approach whereby a liquid crystal tuneable emission filter is employed to sequentially acquire a λ-stack of images across a spectral range, involving illumination and imaging of the entire area, wavelength-at-a-time. This technique has traditionally suffered from speed of acquisition limitations but is boosted by EMCCD sensitivity and readout speed advantages.
Excitation spectroscopic imaging can also be carried out, by scanning a tuneable wavelength excitation source, analyzing the resultant emission intensity. Spectral unmixing algorithms can be applied just as readily to data acquired in this fashion.






 Line scan spectral imaging
Line scan spectral imaging