Here we define Field of View (FOV) by detector size and microscope objective, and Field of Illumination (FOI) relative to the detector and in the image plane.
In a microscope system, the camera is coupled via a C-mount adapter and located in a primary image plane (PIP). The PIP is our reference for the definition of FOV and FOI.
In the PIP, FOV is defined as the extent of the image sensor in X and Y dimensions. For consistency we define FOI as the extent of illumination in the PIP. This is convenient because we can easily calculate overlap between the two.
As mentioned above, it is common to match FOV and FOI, but with active illumination other factors such as Power Density (PD) or Resolution may also be important considerations. Mosaic is equipped a 2X zoom laser collimator so you can trade FOI for PD.
In a fluorescence microscope resolution is dominated by the objective lens which both illuminates and images the specimen. The objective numerical aperture (NA) and the wavelength of detected light (l) define Resolution, RXY by the Rayleigh criterion as follows:
RXY = 0.61 * l/NA;
In the table below RXY is computed for a range of objective lenses with l = 500 nm.
| Magnification (immersion medium) |
Numerical Aperture |
RXY (µm) |
| 20 (air) |
0.6 |
0.51 |
| 40 (air) |
0.8 |
0.38 |
| 40 (WI) |
1.0 |
0.30 |
| 60 (WI) |
1.1 |
0.28 |
| 60 (Oil) |
1.4 |
0.22 |
| 100 (Oil) |
1.4 |
0.22 |
1/RXY is a good approximation of the maximum spatial frequency in the image. To capture all information in the image (e.g. with a CCD detector) we must sample at frequency F to avoid “aliasing errors”. This is known as the Nyquist criterion:
Sampling frequency, F = 2/(RXY)
Translating into the PIP with objective magnification of MO, we can compute the sensor pixel size required to fulfill the Nyquist criterion:
2 * Px = MO * RXY
Subject to application requirements, Nyquist may or may not be necessary, Using an objective lens of 100X, 1.4 NA we see that Neo, Clara, iXon3 885 and Luca R are all capable of achieving the Nyquist criterion: 2 * Px = 22 µm. While at 60X 1.4 NA, only Neo and Clara can provide small enough pixels.
MicroPoint and FRAPPA use Gaussian laser beams and Gaussian beams remain Gaussian with a theoretical minimum focus spot diameter of l, where l is the wavelength. This can only be achieved if the collimated laser beam fills the objective aperture.
The following variation on the Rayleigh criterion provides a definition for spatial resolution SXY of the illumination system:
SXY = 1.22 lf/D; f = focal length of objective,
D is beam diameter.
This is the size of the smallest object the microscope can resolve, sometimes called the diffraction limit, and is also the diameter of the smallest spot to which a collimated beam of incoherent light can be focused. The shape of the spot is an Airy disk or optical point spread function, PSF, characteristic of the system.
| Magnification (immersion medium) |
Numerical Aperture |
SXY (µm) |
| 20 (air) |
0.6 |
1.02 |
| 40 (air) |
0.8 |
0.76 |
| 40 (WI) |
1.0 |
0.6 |
| 60 (WI) |
1.1 |
0.56 |
| 60 (Oil) |
1.4 |
0.44 |
| 100 (Oil) |
1.4 |
0.40 |
The table above shows the approximate theoretical smallest spot sizes for a perfect microscope.
Power density, PD in the specimen plane is estimated from the ratio of beam power and area. Spectral transmission and chromatic errors in the microscope objective are critical to performance. To estimate specimen plane PDS, the Mosaic output beam power density is multiplied by the square of the magnification and the systems spectral transmission, T(l).
PDS = PDD* MO2 * T (l)
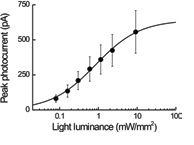
Channelrhodopsin2 (ChRh2) is a light activated cation channel which can be expressed in neurons and used to control behavior in host organisms, including mice, c. elegans and drosophila. Stimulation with blue light (~470 nm), the power density, for photo-activation of ChRh2 is in the range 0.1-10 mW mm-2 and has a wide dynamic range.
Explore our related assets below...


